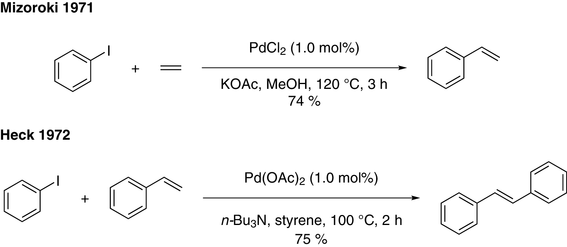
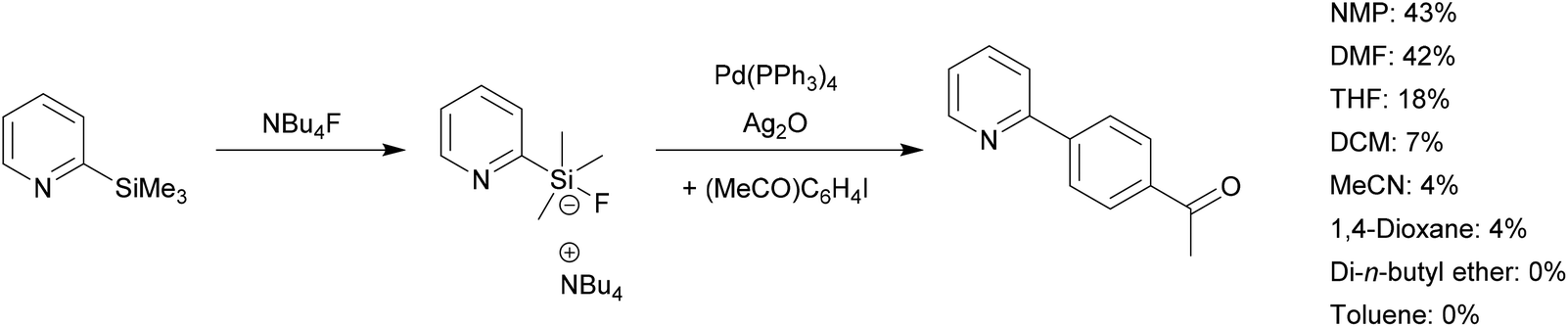
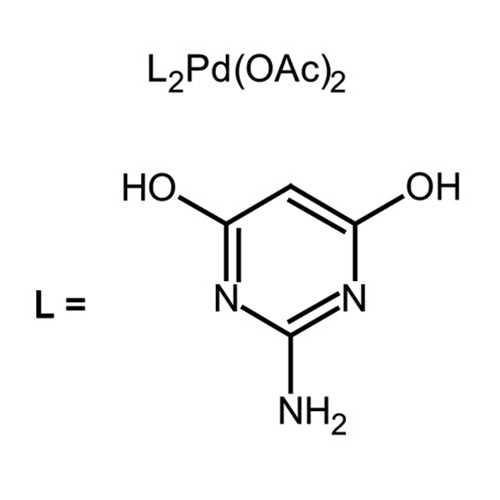
Recent Advances in Pd‐Catalyzed Cross‐Coupling Reaction in Ionic Liquids - Li - 2018 - European Journal of Organic Chemistry - Wiley Online Library

Recent Progress in the Use of Pd-Catalyzed C-C Cross-Coupling Reactions in the Synthesis of Pharmaceutical Compounds
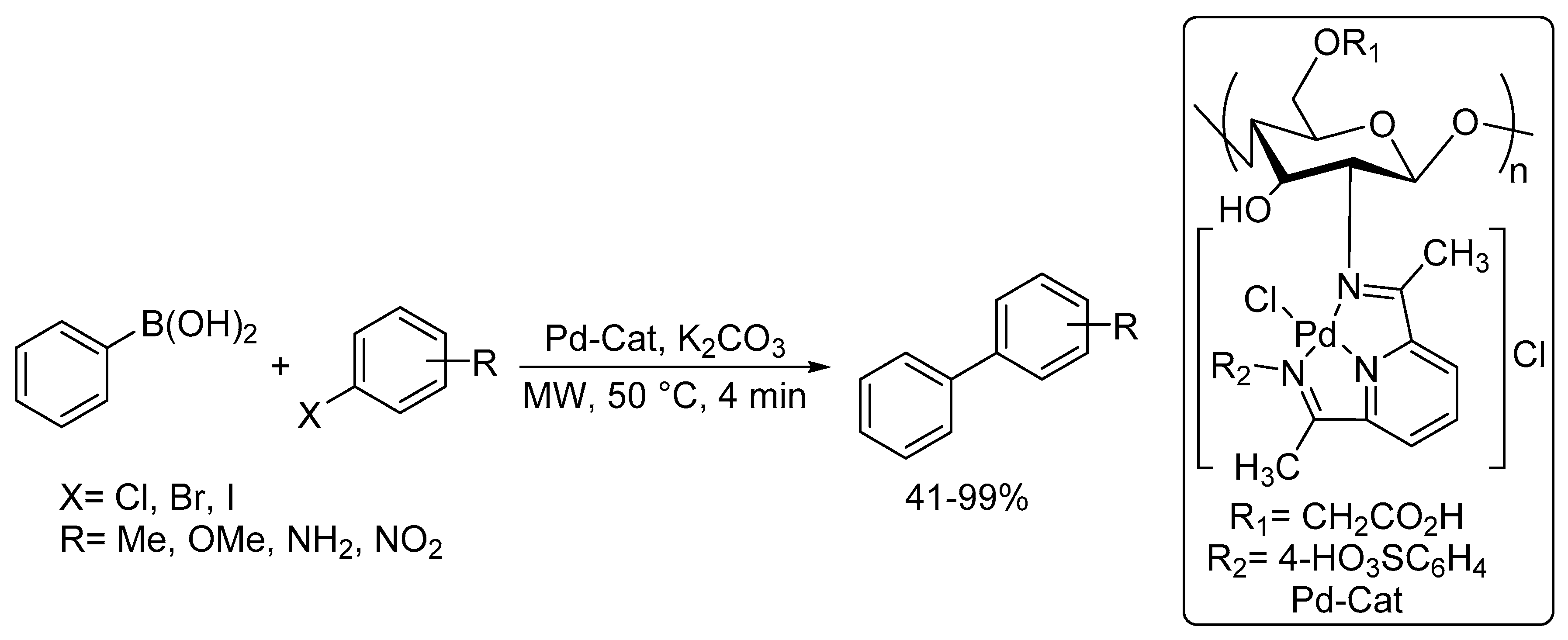
Catalysts | Free Full-Text | Microwave-Assisted Palladium-Catalyzed Cross-Coupling Reactions: Generation of Carbon–Carbon Bond | HTML

Simple aminobenzoic acid promoted palladium catalyzed room temperature Suzuki–Miyaura cross-coupling reaction in aqueous media - ScienceDirect
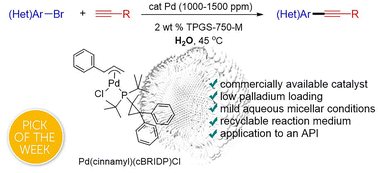
ppm Pd-catalyzed, Cu-free Sonogashira couplings in water using commercially available catalyst precursors - Chemical Science (RSC Publishing)

Biological & Organometallic Catalysis Laboratories - An efficient protocol for the palladium-catalysed Suzuki-Miyaura cross-coupling
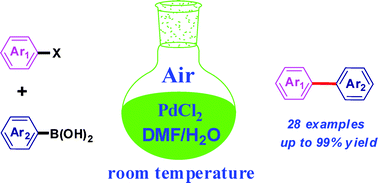
A simple and efficient protocol for a palladium-catalyzed ligand-free Suzuki reaction at room temperature in aqueous DMF - Green Chemistry (RSC Publishing)
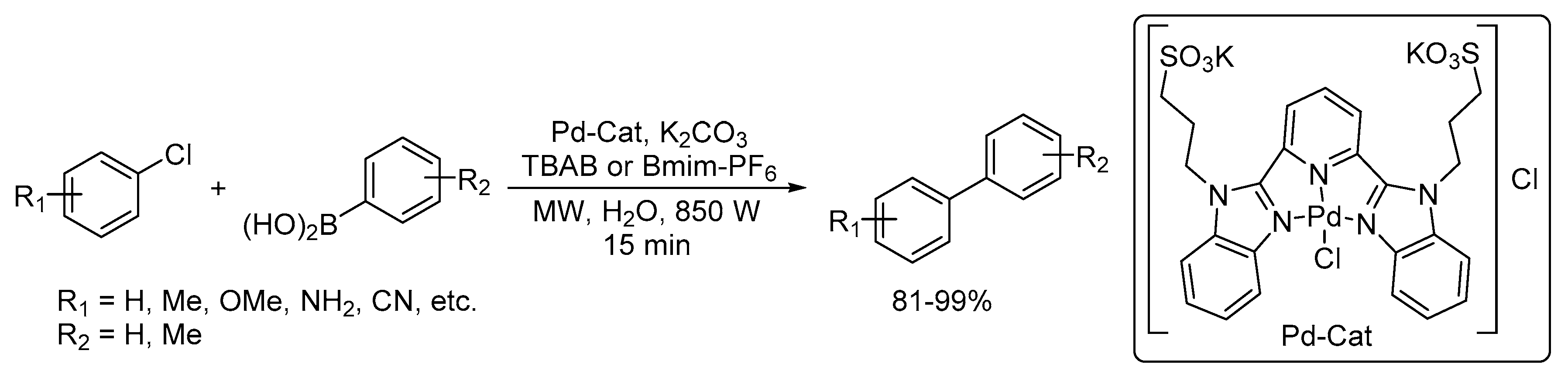
Catalysts | Free Full-Text | Microwave-Assisted Palladium-Catalyzed Cross-Coupling Reactions: Generation of Carbon–Carbon Bond | HTML

Stereoselectivity in Pd-catalysed cross-coupling reactions of enantioenriched nucleophiles | Nature Reviews Chemistry

Water-soluble SNS cationic palladium(II) complexes and their Suzuki–Miyaura cross-coupling reactions in aqueous medium

Combination of a Suzuki cross-coupling reaction using a water-soluble palladium catalyst with an asymmetric enzymatic reduction towards a one-pot process in aqueous medium at room temperature - ScienceDirect

Frontiers | Palladium Mesoionic Carbene Pre-catalyst for General Cross-Coupling Transformations in Deep Eutectic Solvents | Chemistry
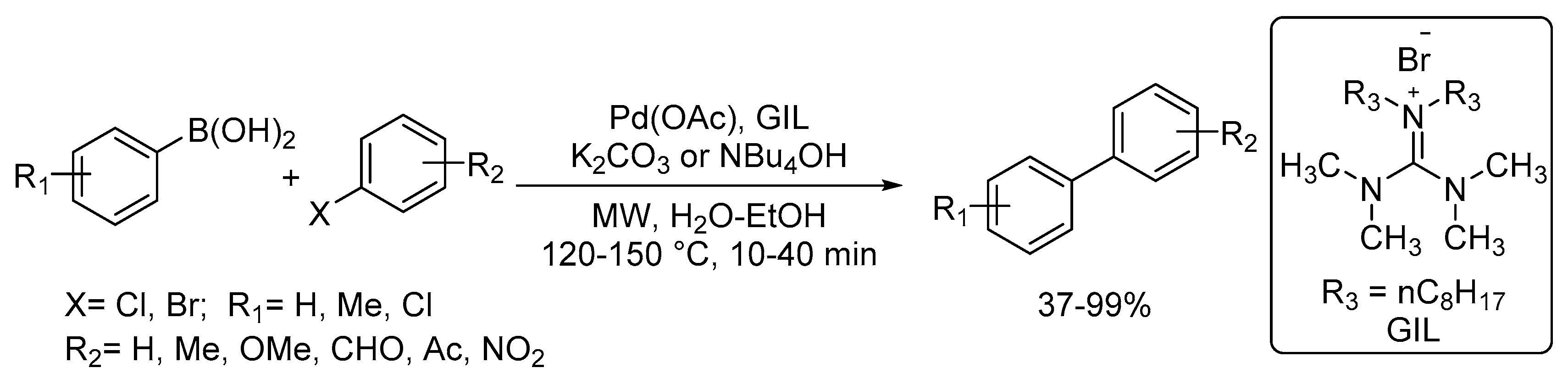
Catalysts | Free Full-Text | Microwave-Assisted Palladium-Catalyzed Cross-Coupling Reactions: Generation of Carbon–Carbon Bond | HTML
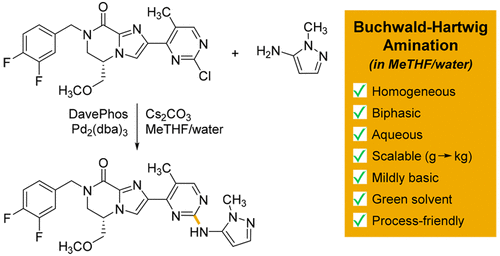
Biphasic Aqueous Reaction Conditions for Process-Friendly Palladium- Catalyzed C–N Cross-Coupling of Aryl Amines - Org. Process Res. Dev. - X-MOL

Ligand‐free Palladium‐Catalyzed Carbonylative Suzuki Coupling of Aryl Iodides in Aqueous CH3CN with Sub‐stoichiometric Amount of Mo(CO)6 as CO Source - Sun - 2019 - Advanced Synthesis & Catalysis - Wiley Online Library

Studies on Pd/NiFe 2 O 4 catalyzed ligand-free Suzuki reaction in aqueous phase: synthesis of biaryls, terphenyls and polyaryls – topic of research paper in Chemical sciences. Download scholarly article PDF and