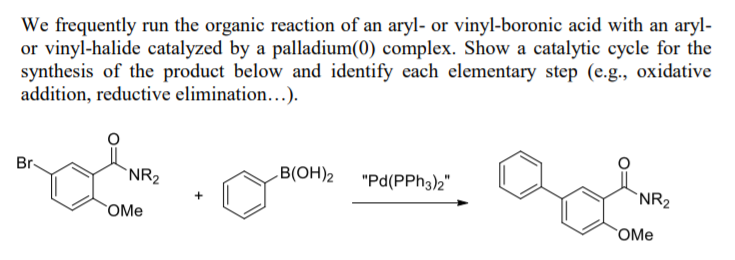
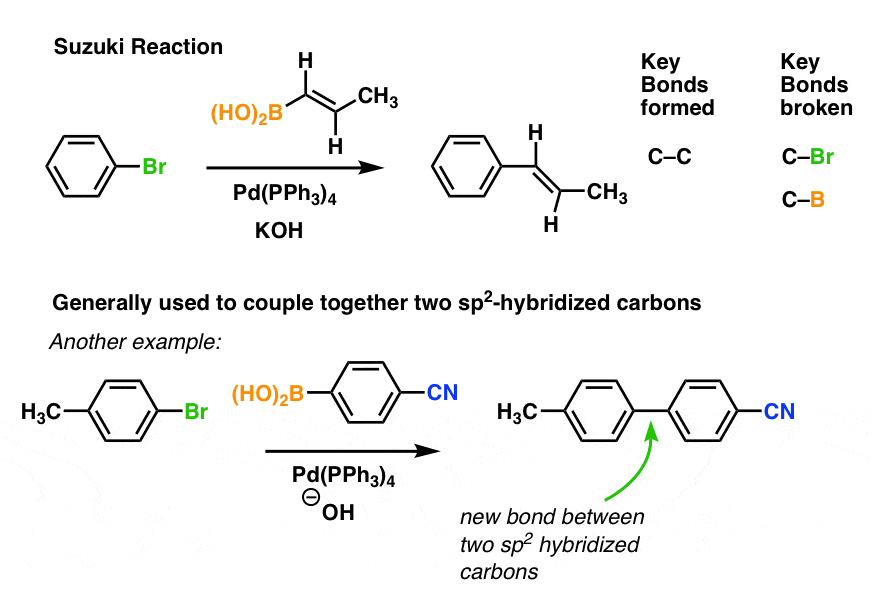
PLOS ONE: Supported Palladium Nanoparticles Synthesized by Living Plants as a Catalyst for Suzuki-Miyaura Reactions

Supported Palladium Nanoparticles that Catalyze Aminocarbonylation of Aryl Halides with Amines using Oxalic Acid as a Sustainable CO Source,Chemistry - A European Journal - X-MOL
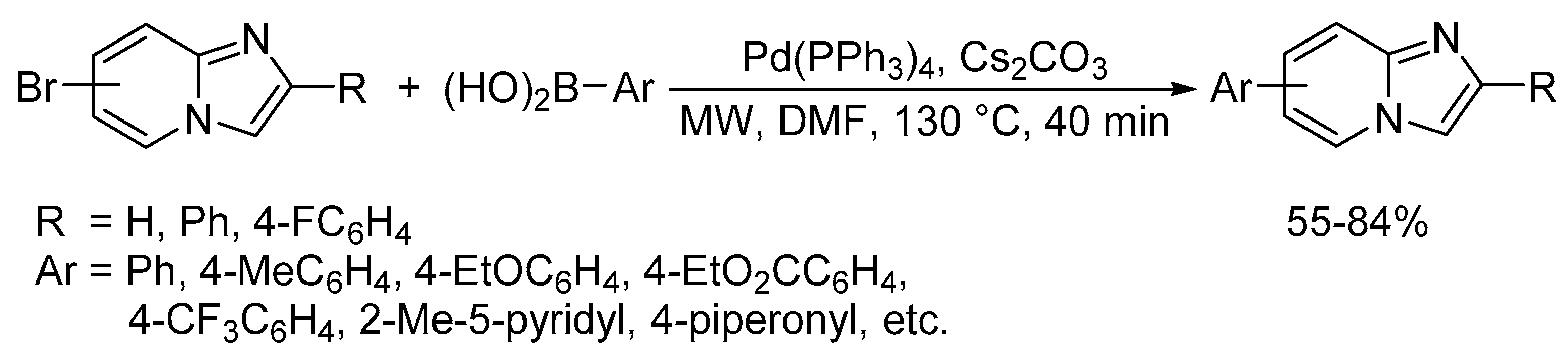
Catalysts | Free Full-Text | Microwave-Assisted Palladium-Catalyzed Cross-Coupling Reactions: Generation of Carbon–Carbon Bond | HTML
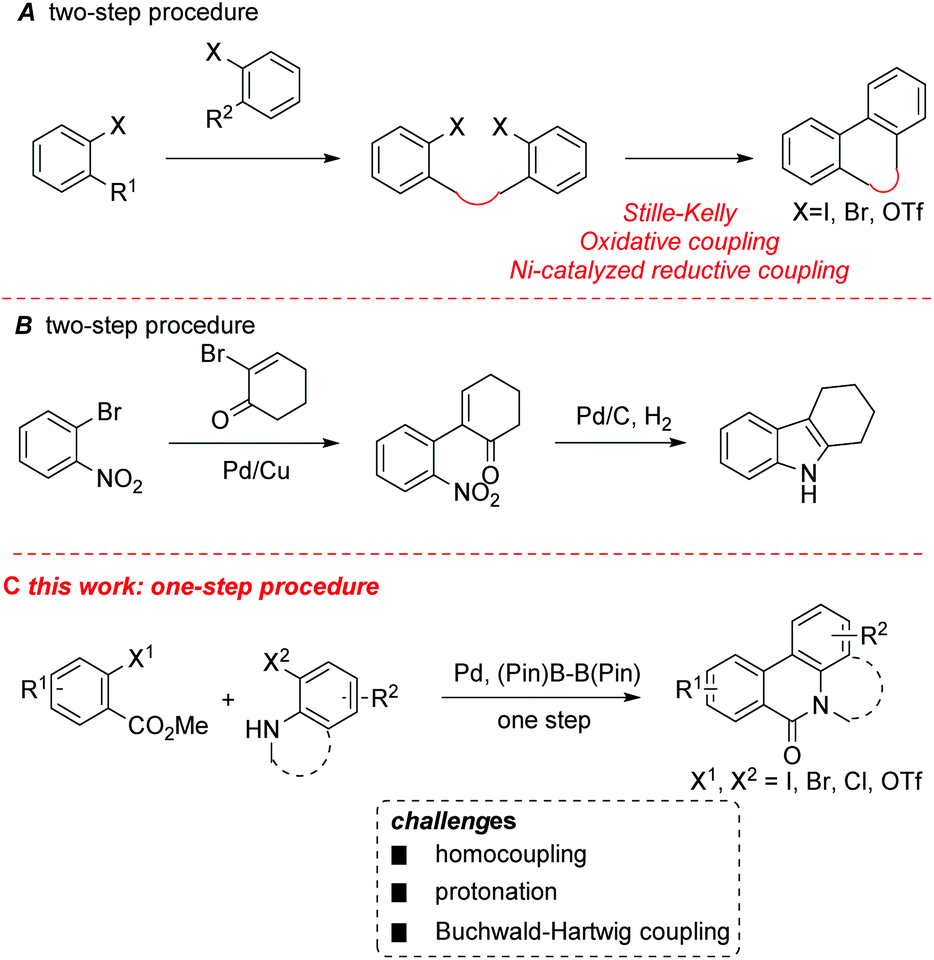
A Pd-catalyzed, boron ester-mediated, reductive cross-coupling of two aryl halides to synthesize tricyclic biaryls - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C7OB01237C
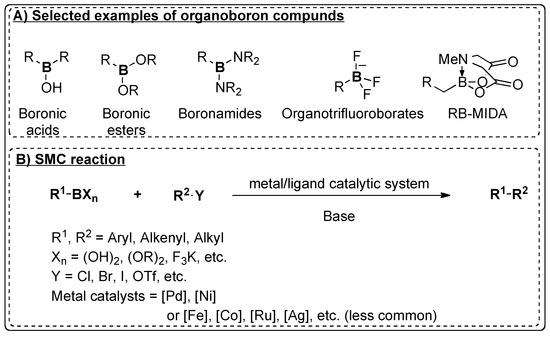
Catalysts | Free Full-Text | Recent Advances in Metal-Catalyzed Alkyl–Boron (C(sp3)–C(sp2)) Suzuki-Miyaura Cross-Couplings | HTML

Figure 1 from Palladium-Catalyzed Synthesis of (Hetero)Aryl Alkyl Sulfones from (Hetero)Aryl Boronic Acids, Unactivated Alkyl Halides, and Potassium Metabisulfite. | Semantic Scholar

Palladium-catalyzed cross-coupling reactions of aryl boronic acids with aryl halides in water - PDF Free Download

Table 1 from Fibrous nano-silica (KCC-1)-supported palladium catalyst: Suzuki coupling reactions under sustainable conditions. | Semantic Scholar

Palladium-catalyzed cross-coupling reactions of aryl boronic acids with aryl halides in water - PDF Free Download

Studies on Pd/NiFe 2 O 4 catalyzed ligand-free Suzuki reaction in aqueous phase: synthesis of biaryls, terphenyls and polyaryls – topic of research paper in Chemical sciences. Download scholarly article PDF and
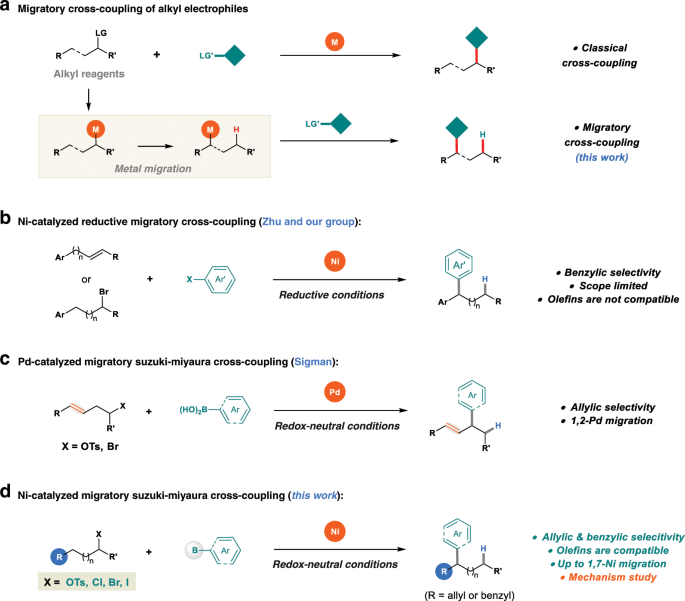
Reaction scope and mechanistic insights of nickel-catalyzed migratory Suzuki–Miyaura cross-coupling | Nature Communications
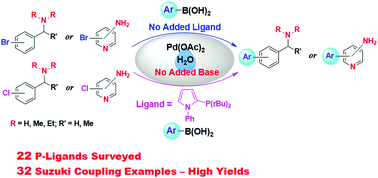
Pd-Catalyzed Suzuki coupling reactions of aryl halides containing basic nitrogen centers with arylboronic acids in water in the absence of added base - New Journal of Chemistry (RSC Publishing)
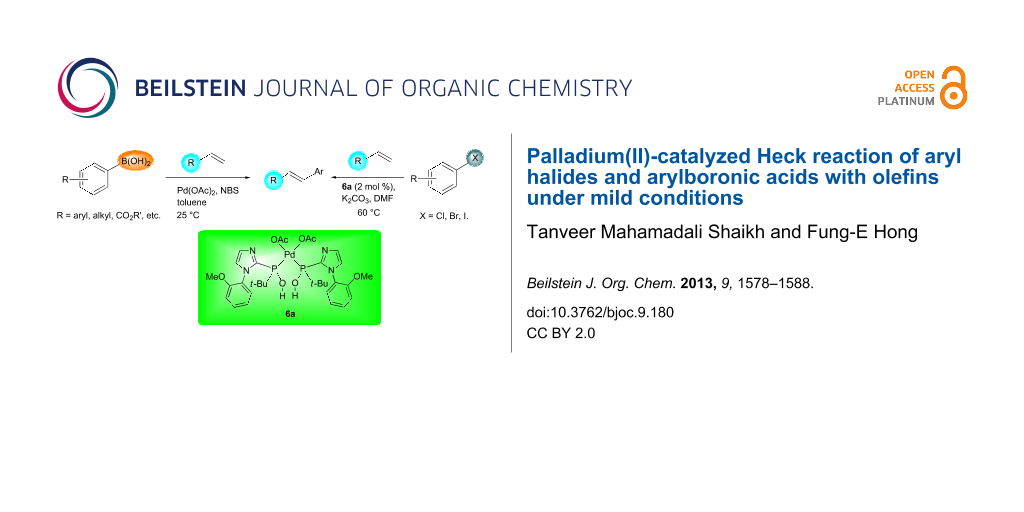
Palladium(II)-catalyzed Heck reaction of aryl halides and arylboronic acids with olefins under mild conditions

Palladium‐Catalyzed Direct Arylation of (Hetero)Arenes with Aryl Boronic Acids - Yang - 2008 - Angewandte Chemie International Edition - Wiley Online Library
![Palladium-catalyzed Suzuki cross-coupling of aryl halides with aryl boronic acids in the presence of glucosamine-based phosphines - [PDF Document] Palladium-catalyzed Suzuki cross-coupling of aryl halides with aryl boronic acids in the presence of glucosamine-based phosphines - [PDF Document]](https://demo.fdocuments.in/img/742x1000/reader018/reader/2020012214/575021491a28ab877e9f0ef3/r-2.jpg?t=1595669709)
Palladium-catalyzed Suzuki cross-coupling of aryl halides with aryl boronic acids in the presence of glucosamine-based phosphines - [PDF Document]
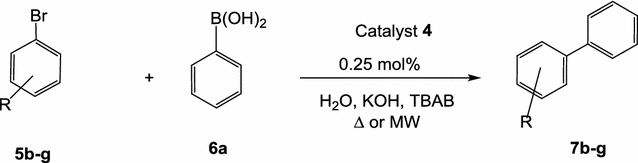
Novel pyridine-based Pd(II)-complex for efficient Suzuki coupling of aryl halides under microwaves irradiation in water | SpringerLink

Suzuki-Miyaura cross-coupling of phenylboronic acid with aryl halides catalyzed by palladium and nickel species supported on alumina-based oxides - ScienceDirect

i>N</i>-Doped porous carbon supported palladium nanoparticles as a highly efficient and recyclable catalyst for the Suzuki coupling reaction