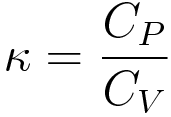![PDF] Worldwide Assessment of Industry Leak Detection Capabilities for Single & Multiphase Pipelines | Semantic Scholar PDF] Worldwide Assessment of Industry Leak Detection Capabilities for Single & Multiphase Pipelines | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/2dbc9552819c5dbc5eb1dbc882278fc7a0eaa45b/83-Figure7.3-1.png)
PDF] Worldwide Assessment of Industry Leak Detection Capabilities for Single & Multiphase Pipelines | Semantic Scholar

Cp-Cv for real gas, in terms of alpha & beta, change in internal energy with respect to volume - YouTube

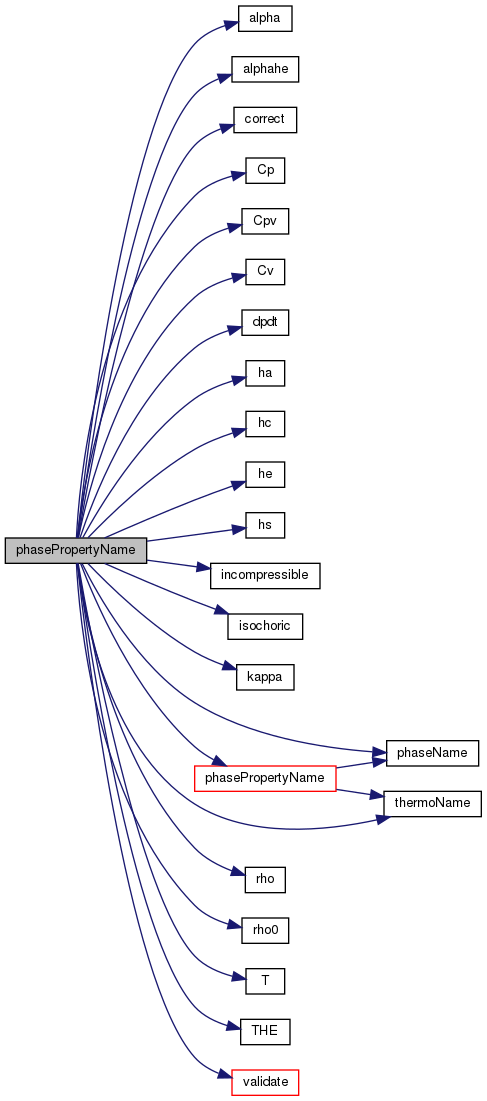
A class diagram of (a) the thermophysicalModels library with critical... | Download Scientific Diagram

Energy Conversion CHE 450/550. Ideal Gas Basics and Heat Capacities - I Ideal gas: – a theoretical gas composed of a set of non-interacting point particles. - ppt download