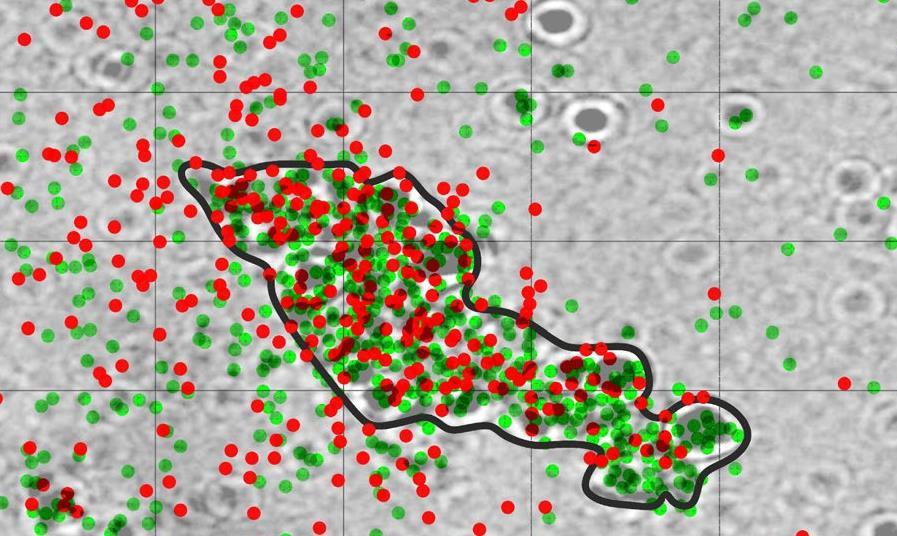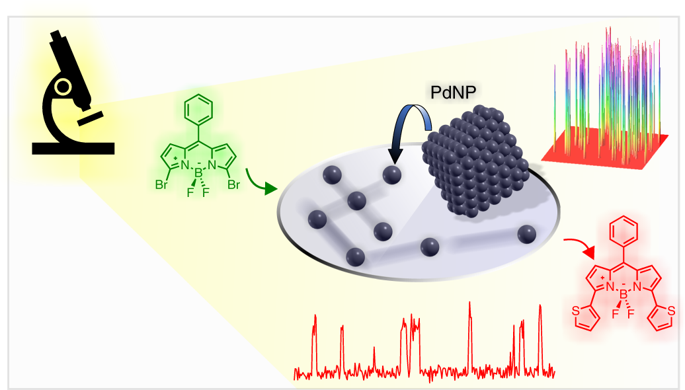
Real-time fluorescence imaging of a heterogeneously catalysed Suzuki–Miyaura reaction - Nat. Catal. - X-MOL
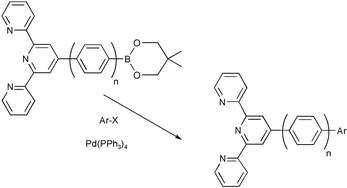
The synthesis of 4′-aryl substituted terpyridines by Suzuki cross-coupling reactions: substituent effects on ligand fluorescence - Journal of the Chemical Society, Perkin Transactions 2 (RSC Publishing)

Scheme 6. Synthesis of fluorescent nucleosides following Suzuki-Miyuara... | Download Scientific Diagram

Palladium-Catalyzed Suzuki-Miyaura Cross-coupling Reactions Employing Dialkylbiaryl Phosphine Ligands | Scinapse
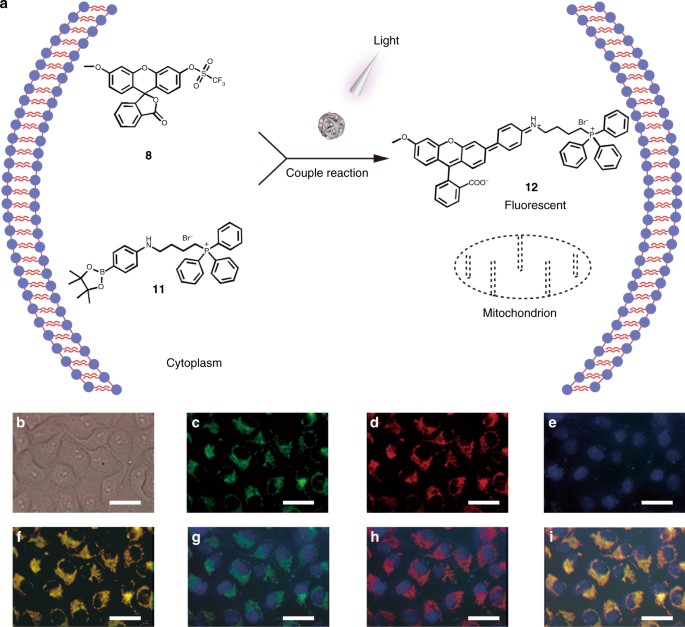
Designed heterogeneous palladium catalysts for reversible light-controlled bioorthogonal catalysis in living cells | Nature Communications

Nanosized palladium-catalyzed Suzuki–Miyaura coupling polymerization: synthesis of soluble aromatic poly(ether ketone)s | Polymer Journal

Analysis of the Suzuki–Miyaura cross-coupling reaction a, SMCC reaction... | Download Scientific Diagram

Figure 2 from A fluorescence active catalyst support comprising carbon quantum dots and magnesium oxide doping for stabilization of palladium nanoparticles: Application as a recoverable catalyst for Suzuki reaction in water

Post-transcriptional labeling by using Suzuki-Miyaura cross-coupling generates functional RNA probes. - Abstract - Europe PMC

Evidence for the Surface‐Catalyzed Suzuki–Miyaura Reaction over Palladium Nanoparticles: An Operando XAS Study - Ellis - 2010 - Angewandte Chemie International Edition - Wiley Online Library

Studies on Pd/NiFe 2 O 4 catalyzed ligand-free Suzuki reaction in aqueous phase: synthesis of biaryls, terphenyls and polyaryls – topic of research paper in Chemical sciences. Download scholarly article PDF and

Front Cover: Recombinant Peptide Fusion Protein‐Templated Palladium Nanoparticles for Suzuki‐Miyaura and Stille Coupling Reactions (ChemCatChem 11/2020) - Mosleh - 2020 - ChemCatChem - Wiley Online Library

A reusable heterogeneous catalyst without leaking palladium for highly-efficient Suzuki–Miyaura reaction in pure water under air† - RSC Adv. - X-MOL

Figure 4 from A fluorescence active catalyst support comprising carbon quantum dots and magnesium oxide doping for stabilization of palladium nanoparticles: Application as a recoverable catalyst for Suzuki reaction in water

Supramolecular photocatalyst of Palladium (II) Encapsulated within Dendrimer on TiO2 nanoparticles for Photo‐induced Suzuki‐Miyaura and Sonogashira Cross‐Coupling reactions - Eskandari - 2019 - Applied Organometallic Chemistry - Wiley Online Library

The synthesis of novel directly conjugated zinc(II) phthalocyanine via palladium-catalyzed Suzuki–Miyaura cross-coupling reaction and its quaternized water-soluble derivative: Investigation of photophysical and photochemical properties - ScienceDirect

Figure 1 from A fluorescence active catalyst support comprising carbon quantum dots and magnesium oxide doping for stabilization of palladium nanoparticles: Application as a recoverable catalyst for Suzuki reaction in water
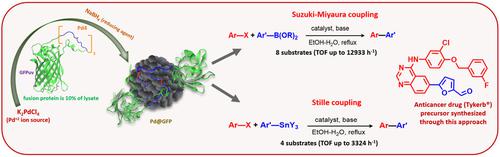
Recombinant Peptide Fusion Protein‐Templated Palladium Nanoparticles for Suzuki‐Miyaura and Stille Coupling Reactions - ChemCatChem - X-MOL
![Synthesis of Fluorescent 2-Substituted 6-(Het)aryl-7-deazapurine Bases {4-(Het)aryl-pyrrolo[2,3-d]pyrimidines} by Aqueous Suzuki–Miyaura Cross-Coupling Reactions - Synthesis - X-MOL Synthesis of Fluorescent 2-Substituted 6-(Het)aryl-7-deazapurine Bases {4-(Het)aryl-pyrrolo[2,3-d]pyrimidines} by Aqueous Suzuki–Miyaura Cross-Coupling Reactions - Synthesis - X-MOL](https://xpic.x-mol.com/thesis%2FSynthesis%2Fi_z0663_ga_10-1055_s-0035-1561287.gif)
Synthesis of Fluorescent 2-Substituted 6-(Het)aryl-7-deazapurine Bases {4-(Het)aryl-pyrrolo[2,3-d]pyrimidines} by Aqueous Suzuki–Miyaura Cross-Coupling Reactions - Synthesis - X-MOL