Palladium-catalyzed asymmetric dearomative alkenylation of indoles through a reductive-Heck reaction - Organic Chemistry Frontiers (RSC Publishing)
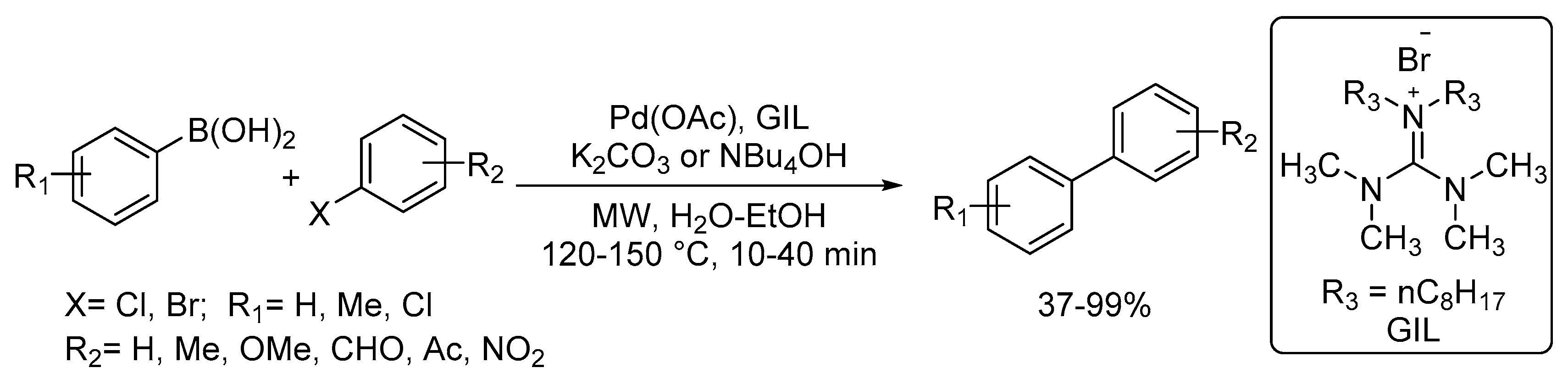
Catalysts | Free Full-Text | Microwave-Assisted Palladium-Catalyzed Cross-Coupling Reactions: Generation of Carbon–Carbon Bond | HTML

Optimization of palladium-catalyzed Ferrier-type C-glycosylation with... | Download Scientific Diagram

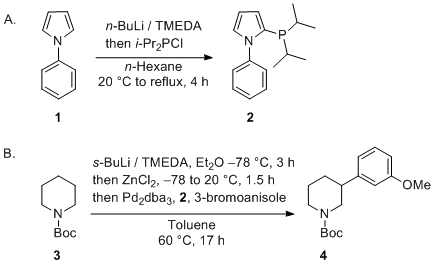
Hydrodehalogenation of halogenated pyridines and quinolines by sodium borohydride/N,N,N′,N′-tetramethylethylenediamine under palladium catalysis
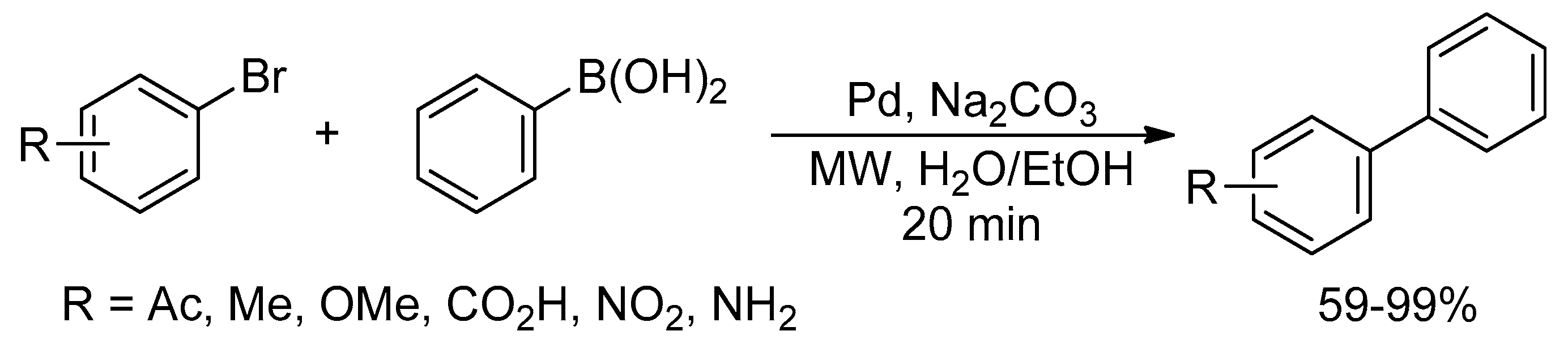
Catalysts | Free Full-Text | Microwave-Assisted Palladium-Catalyzed Cross-Coupling Reactions: Generation of Carbon–Carbon Bond | HTML

One-pot synthesis of self-assembled heteroleptic palladium(II) complexes with tmeda: An application of ligand exchange reactions - ScienceDirect

Palladium-catalyzed decarboxylative gem-selective addition of alkynoic acids to terminal alkynes - Organic Chemistry Frontiers (RSC Publishing)

EP2107047A1 - Method for producing organic compounds by means of a transition metal-catalysed cross-coupling reaction of an aryl-X, heteroaryl-X, cycloalkenyl-X or alkenyl-X compound with an alkyl, alkenyl, cycloalkyl or cycloalkenyl halogenide -

Synthesis of Internal Alkynes by Pd(PPh3)4/TMEDA‐Catalyzed Kumada Cross‐Coupling of Alkynyl Halides with Grignard Reagents - Zhang - 2014 - European Journal of Organic Chemistry - Wiley Online Library

Table 1 from An efficient diamine.copper complex-catalyzed coupling of arylboronic acids with imidazoles | Semantic Scholar

Figure 1 from Palladium-catalyzed enantioselective alpha-arylation and alpha-vinylation of oxindoles facilitated by an axially chiral P-stereogenic ligand. | Semantic Scholar

Pd-catalyzed synthesis of α,β-unsaturated ketones by carbonylation of vinyl triflates and nonaflates - Chemical Communications (RSC Publishing)

NaBH4-TMEDA and a palladium catalyst as efficient regio- and chemoselective system for the hydrodehalogenation of halogenated heterocycles - ScienceDirect

Regioselective and Stereospecific Cross‐Coupling of Primary Allylic Amines with Boronic Acids and Boronates through Palladium‐Catalyzed CN Bond Cleavage - Li - 2012 - Angewandte Chemie International Edition - Wiley Online Library
![Combined directed ortho zincation and palladiumcatalyzed strategies: Synthesis of 4,n dimethoxysubstituted benzo[b]furans Combined directed ortho zincation and palladiumcatalyzed strategies: Synthesis of 4,n dimethoxysubstituted benzo[b]furans](https://data02.123dok.com/thumb/yd/76/9oly/Pki4ZiPT5VIqTD1so/cover.webp)
Combined directed ortho zincation and palladiumcatalyzed strategies: Synthesis of 4,n dimethoxysubstituted benzo[b]furans

Palladium(II)-catalyzed C-H activation/C-C cross-coupling reactions: versatility and practicality. - Abstract - Europe PMC