Facile and selective cleavage of allyl ethers, amines and esters using polymethylhydrosiloxane-ZnCl2/Pd(PPh3)4

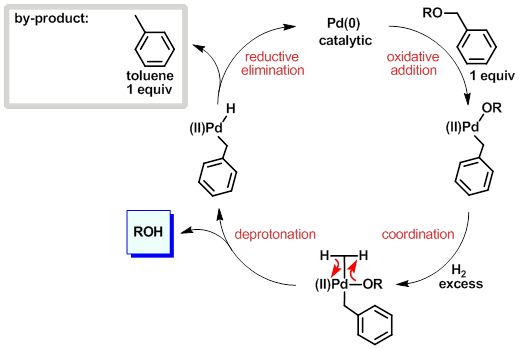
Palladium-catalyzed reaction of tributyltin hydride. Selective and very mild deprotection of allyl and allyloxycarbonyl derivatives of amino-acids
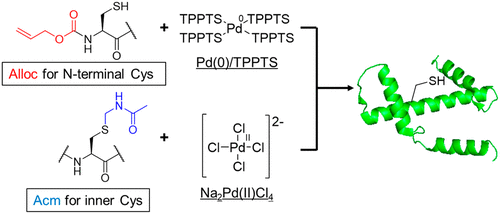
Chemical Synthesis of Cys-Containing Protein via Chemoselective Deprotection with Different Palladium Complexes.,Organic Letters - X-MOL

Facile and selective cleavage of allyl ethers, amines and esters using polymethylhydrosiloxane–ZnCl2/Pd(PPh3)4 - ScienceDirect

Intracellular Deprotection Reactions Mediated by Palladium Complexes Equipped with Designed Phosphine Ligands. - ACS Catal. - X-MOL

Figure 2 from Palladium-triggered deprotection chemistry for protein activation in living cells. | Semantic Scholar

Palladium-triggered deprotection chemistry for protein activation in living cells | Nature Chemistry
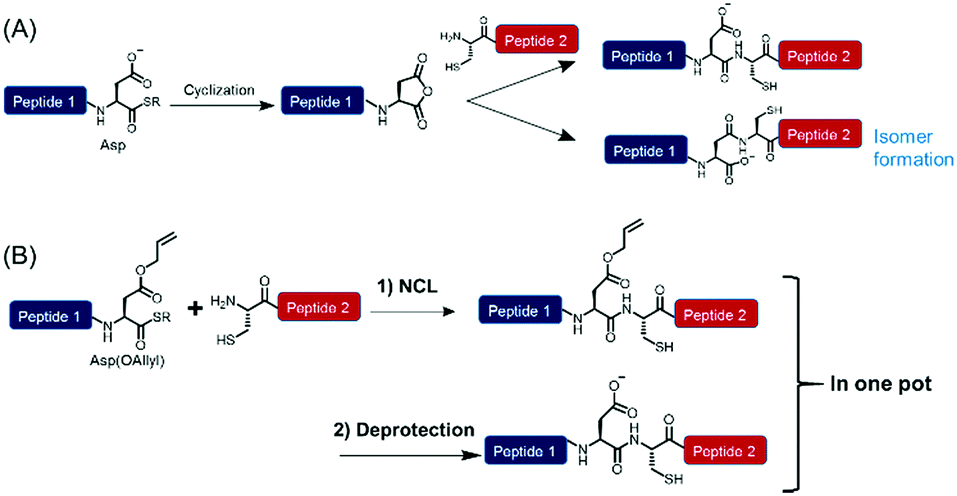
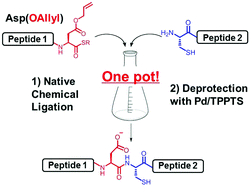
Efficient peptide ligation between allyl-protected Asp and Cys followed by palladium-mediated deprotection - Chemical Communications (RSC Publishing) DOI:10.1039/C8CC01965G

Palladium prompted on-demand cysteine chemistry for the synthesis of challenging and uniquely modified proteins | Nature Communications

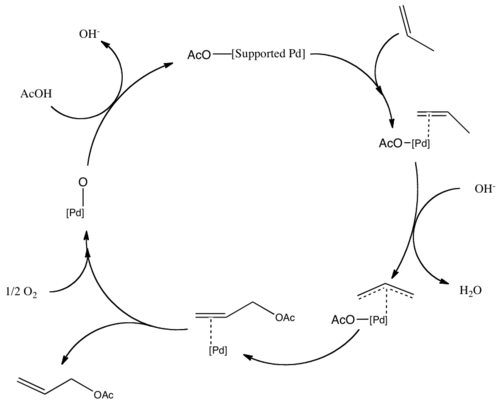
Pd(II)-catalyzed deprotection of acetals and ketals containing acid sensitive functional groups - ScienceDirect
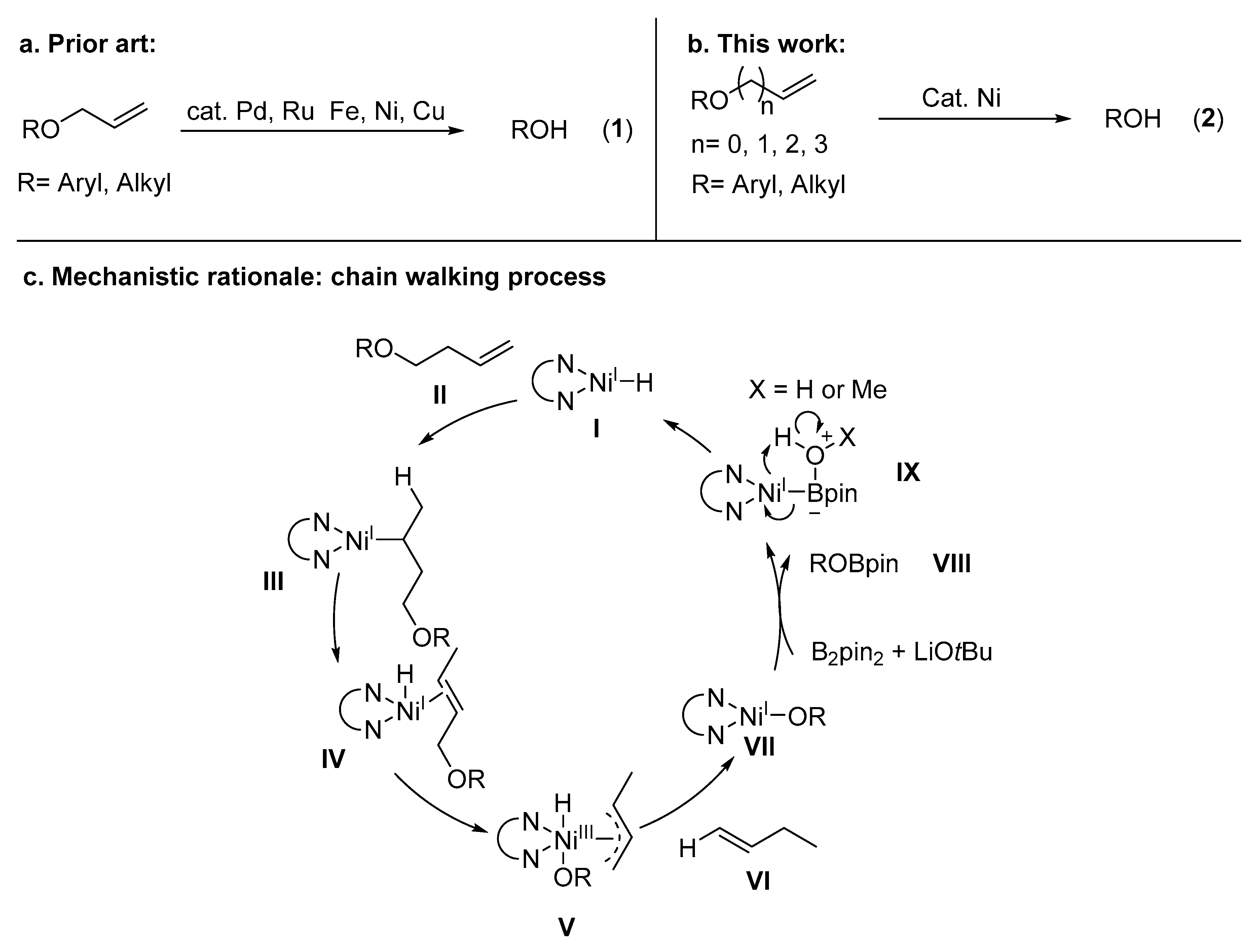
Molecules | Free Full-Text | Nickel-Catalyzed Removal of Alkene Protecting Group of Phenols, Alcohols via Chain Walking Process | HTML

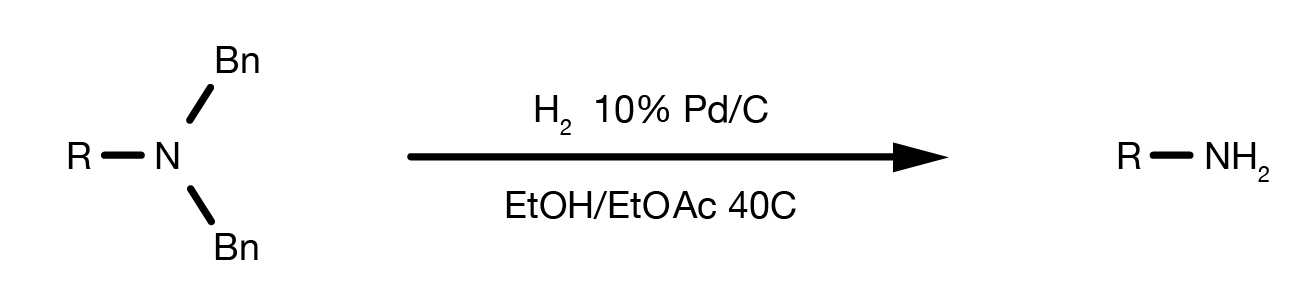
Deprotection reactions of a genetically encoded caged lysine with the... | Download Scientific Diagram