![Palladium-catalyzed tandem reaction to construct benzo[c]phenanthridine: application to the total synthesis of benzo[c]phenanthridine alkaloids - Organic & Biomolecular Chemistry (RSC Publishing) Palladium-catalyzed tandem reaction to construct benzo[c]phenanthridine: application to the total synthesis of benzo[c]phenanthridine alkaloids - Organic & Biomolecular Chemistry (RSC Publishing)](https://pubs.rsc.org/image/article/2011/ob/c0ob01208d/c0ob01208d-f1.gif)
Palladium-catalyzed tandem reaction to construct benzo[c]phenanthridine: application to the total synthesis of benzo[c]phenanthridine alkaloids - Organic & Biomolecular Chemistry (RSC Publishing)

Synthesis of 6‐Substituted Phenanthridine Derivatives by Palladium‐Catalysed Domino Suzuki–Miyaura/Aza‐Michael Reactions - Bao - 2014 - European Journal of Organic Chemistry - Wiley Online Library
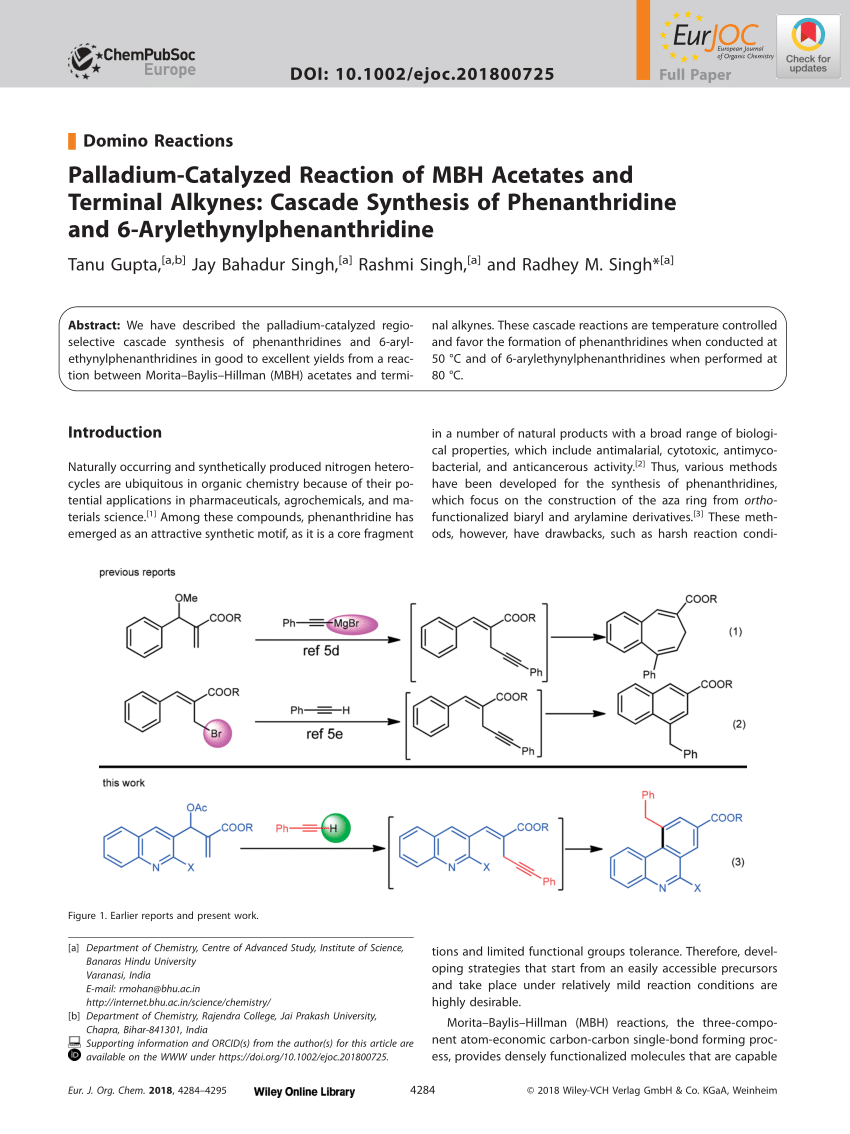
PDF) Palladium-Catalyzed Reaction of MBH Acetate with Terminal Alkyne: Cascade Synthesis of Phenanthridine and 6-Arylethynyl Phenanthridine

PDF) One-Pot Palladium-Catalyzed Synthesis of Selectively Substituted Phenanthridines by Sequential Aryl-Aryl and Heck Couplings, Aza-Michael and Retro-Mannich Reactions | Elena Motti - Academia.edu

Come-back of phenanthridine and phenanthridinium derivatives in the 21st century. - PDF Download Free

Table 2 from Synthesis of phenanthridines via palladium-catalyzed picolinamide-directed sequential C–H functionalization | Semantic Scholar
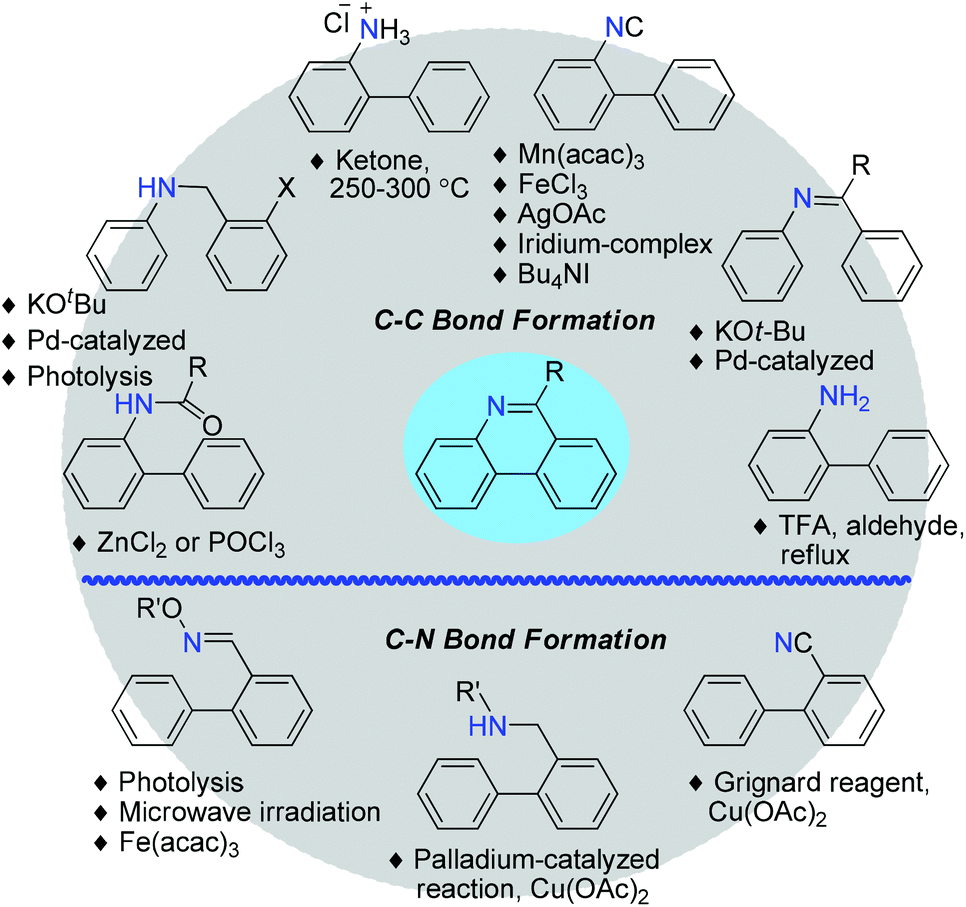
A new synthetic approach to 6-unsubstituted phenanthridine and phenanthridine-like compounds under mild and metal-free conditions - Organic & Biomolecular Chemistry (RSC Publishing)

Palladium-catalyzed intramolecular C−H bond functionalization of trifluoroacetimidoyl chloride derivatives: Synthesis of 6-trifluoromethyl- phenanthridines - ScienceDirect

PDF) An Investigation of a Palladium Catalysed Biaryl Synthesis of Pyrrolophenanthridine Derivatives. Extension of the Heck Reaction
![Synthesis of pyrrolo[1,2- f ]phenanthridine-annulated polycyclic heterocycles from palladium-catalyzed intramolecular direct arylation reaction | SpringerLink Synthesis of pyrrolo[1,2- f ]phenanthridine-annulated polycyclic heterocycles from palladium-catalyzed intramolecular direct arylation reaction | SpringerLink](https://media.springernature.com/lw685/springer-static/image/art%3A10.1007%2Fs00706-011-0564-9/MediaObjects/706_2011_564_Sch1_HTML.gif)
Synthesis of pyrrolo[1,2- f ]phenanthridine-annulated polycyclic heterocycles from palladium-catalyzed intramolecular direct arylation reaction | SpringerLink
![Synthesis of Novel Isoquinolino[5,4-ab]phenanthridine Derivatives via Pictet–Spengler Reaction - Synthesis - X-MOL Synthesis of Novel Isoquinolino[5,4-ab]phenanthridine Derivatives via Pictet–Spengler Reaction - Synthesis - X-MOL](https://xpic.x-mol.com/20190227%2F10.1055_s-0037-1610670.jpg)
Synthesis of Novel Isoquinolino[5,4-ab]phenanthridine Derivatives via Pictet–Spengler Reaction - Synthesis - X-MOL
![Palladium‐Catalyzed Synthesis of 6H‐Dibenzo[c,h]chromenes and 5,6‐Dihydrobenzo[c]phenanthridines: Application to the Synthesis of Dibenzo[c,h]chromene‐6‐ones, Benzo[c]phenanthridines, and Arnottin I - Adv. Synth. Catal. - X-MOL Palladium‐Catalyzed Synthesis of 6H‐Dibenzo[c,h]chromenes and 5,6‐Dihydrobenzo[c]phenanthridines: Application to the Synthesis of Dibenzo[c,h]chromene‐6‐ones, Benzo[c]phenanthridines, and Arnottin I - Adv. Synth. Catal. - X-MOL](https://xpic.x-mol.com/20191015%2F10.1002_adsc.201900833.jpg)
Palladium‐Catalyzed Synthesis of 6H‐Dibenzo[c,h]chromenes and 5,6‐Dihydrobenzo[c]phenanthridines: Application to the Synthesis of Dibenzo[c,h]chromene‐6‐ones, Benzo[c]phenanthridines, and Arnottin I - Adv. Synth. Catal. - X-MOL
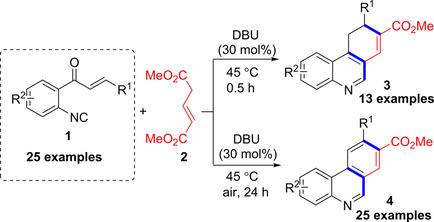
Synthesis and Reactivity of o‐Enoyl Arylisocyanides: Access to Phenanthridine‐8‐Carboxylate Derivatives - Adv. Synth. Catal. - X-MOL
![Palladium-catalyzed arylation of 1,4-naphthoquinones with aryl iodides and its synthetic application to the benzo[b]phenanthridine skeleton - Tetrahedron Lett. - X-MOL Palladium-catalyzed arylation of 1,4-naphthoquinones with aryl iodides and its synthetic application to the benzo[b]phenanthridine skeleton - Tetrahedron Lett. - X-MOL](https://xpic.x-mol.com/20200911%2F10.1016_j.tetlet.2020.152446.jpg)
Palladium-catalyzed arylation of 1,4-naphthoquinones with aryl iodides and its synthetic application to the benzo[b]phenanthridine skeleton - Tetrahedron Lett. - X-MOL

Palladium-Catalyzed Synthesis of Phenanthridine/Benzoxazine-Fused Quinazolinones by Intramolecular C-H Bond Activation. | Semantic Scholar
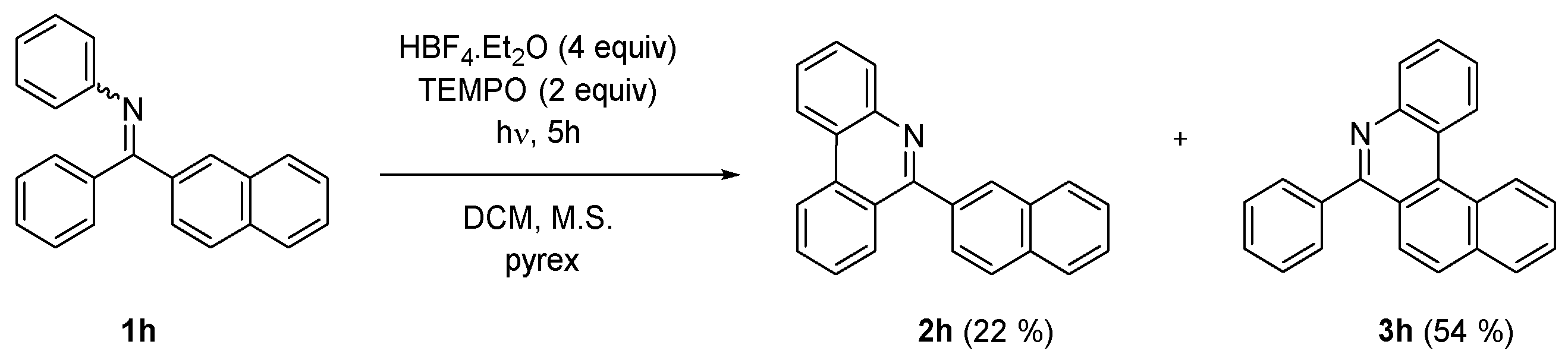
IJMS | Free Full-Text | Oxidative Photocyclization of Aromatic Schiff Bases in Synthesis of Phenanthridines and Other Aza-PAHs | HTML
![Palladium-catalyzed annulation reactions of methyl o-halobenzoates with azabicyclic alkenes: a general protocol for the construction of benzo[c] phenanthridine derivatives - RSC Advances (RSC Publishing) Palladium-catalyzed annulation reactions of methyl o-halobenzoates with azabicyclic alkenes: a general protocol for the construction of benzo[c] phenanthridine derivatives - RSC Advances (RSC Publishing)](https://pubs.rsc.org/en/Content/Image/GA/C3RA42474J)
Palladium-catalyzed annulation reactions of methyl o-halobenzoates with azabicyclic alkenes: a general protocol for the construction of benzo[c] phenanthridine derivatives - RSC Advances (RSC Publishing)

Synthesis of phenanthridines via palladium-catalyzed picolinamide-directed sequential C–H functionalization – topic of research paper in Chemical sciences. Download scholarly article PDF and read for free on CyberLeninka open science hub.
![Straightforward palladium-mediated synthesis and biological evaluation of benzo[j]phenanthridine-7,12-diones as anti-tuberculosis agents. | Semantic Scholar Straightforward palladium-mediated synthesis and biological evaluation of benzo[j]phenanthridine-7,12-diones as anti-tuberculosis agents. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/ad18a5018a64d69d82a3cad2a6a7fcb8983184c3/2-Figure1-1.png)
Straightforward palladium-mediated synthesis and biological evaluation of benzo[j]phenanthridine-7,12-diones as anti-tuberculosis agents. | Semantic Scholar
![Straightforward palladium-mediated synthesis and biological evaluation of benzo[j]phenanthridine-7,12-diones as anti-tuberculosis agents - ScienceDirect Straightforward palladium-mediated synthesis and biological evaluation of benzo[j]phenanthridine-7,12-diones as anti-tuberculosis agents - ScienceDirect](https://ars.els-cdn.com/content/image/1-s2.0-S0223523411008555-fx1.jpg)


![Scheme 1 Retrosynthetic analysis of benzo[c]phenanthridines. | Download Scientific Diagram Scheme 1 Retrosynthetic analysis of benzo[c]phenanthridines. | Download Scientific Diagram](https://www.researchgate.net/profile/Xiao_Xu13/publication/50597439/figure/fig3/AS:669152608010249@1536549822575/Scheme-1-Retrosynthetic-analysis-of-benzocphenanthridines_Q640.jpg)